Ora & Singapore Eye Research Institute: Unparalleled preclinical research for early-stage innovators.
When conceptualizing and commencing the development pipeline, the ability to think ahead to future stages in the clinical pipeline is vital to ensuring the right activities are done at the preclinical phase and avoid costly rework that can set back timelines.
Combined with the Singapore Eye Research Institute’s 30 years of preclinical expertise and world class facilities, our experience and expertise when engaging with early-stage clients can truly allow you to begin your program with the end in mind.
Renowned for its cutting-edge work in the realms of eye disease and visual science, SERI’s research spans basic, clinical, and translational efforts involving experts in ophthalmology, molecular biology, genetics, bioengineering, AI, epidemiology, and population health.
SERI has grown from a founding team of five in 1997 to an integrated organization of more than 240 staff, encompassing clinician scientists, scientists, research fellows, PhD students and support staff. This makes SERI one of the largest research institutes in the Asia Pacific region, publishing an impressive array of more than 5000 scientific papers.
SERI undertakes vision research in collaboration with local clinical ophthalmic centers and biomedical research institutions, as well as major eye centers and research institutes throughout the world. One of the key strengths of SERI is its multidisciplinary, collaborative approach to research that has led to breakthroughs in understanding and treating eye diseases, resulting in innovative diagnostic and therapeutic approaches.
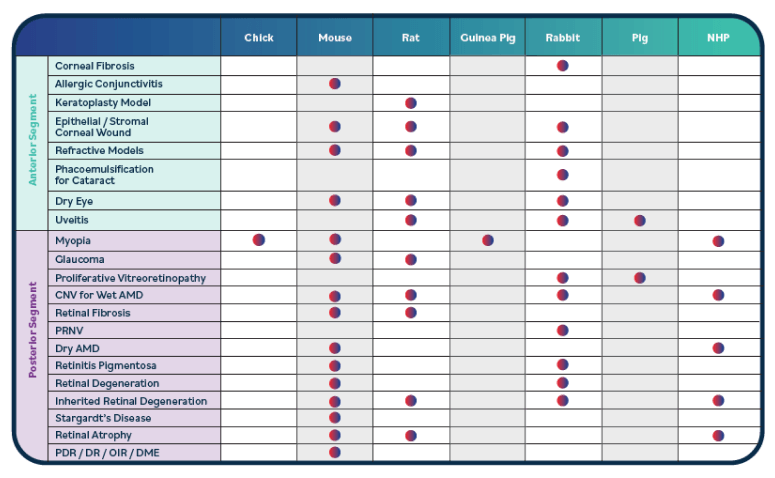
The first steps in any journey are crucial. Ora’s decades of experience in ophthalmic development can ensure that you begin your program with the end in mind.
Ora’s experts, combined with world class full turnkey preclinical facilities at SERI, can help you de-risk your entire clinical program by considering your regulatory, clinical, and investment strategies when designing your preclinical phase.
Regulatory
From designing your preclinical programming to interpreting data and presenting to regulatory agencies.
Investment
Finding the right data points to de-risk your investment strategy.
Clinical
Liaising with Ora’s Therapeutic Area Experts to enable a seamless transition into the clinic.
Leading the charge for this collaboration is Buffie Kerstetter, Director of Preclinical Partnerships, a tenured preclinical business development advocate for Ora’s valued sponsors in preclinical development. Prior to working at Ora Buffie has spent her entire 30-year career working with animals in research as a certified lab animal technologist and providing oversight of animal programs and staff at several large institutions.

Director, Preclinical Partnerships
“We are excited to embark on this pivotal partnership with Ora. This collaboration propels us toward realizing SERI’s vision of becoming an international center of excellence in eye and vision research. Our SERI scientists rank among the top 2% globally in ophthalmology, and our unprecedented impact factor in scientific publications showcase our commitment to advancing eye care. By seamlessly integrating our expertise with Ora’s, we aim to provide unmatched preclinical capabilities, accelerating the development of innovative ophthalmic therapies and reinforcing our position as leaders in translating basic science to clinical applications for sight-threatening disorders.”

Professor Jodhbir Mehta, MD,
Executive director of SERI and deputy chief executive officer (Research), SNEC
Ora, Inc websites use cookies. By continuing to browse the site you are agreeing to our use of cookies.
For more details about cookies and their use, please see our Privacy Policy.